Core Economic Framing
Drug development is fundamentally a probability problem. Traditional non-genetic target discovery pathways can produce roughly a 10% probability of approval from Phase I.
Expected cost per approved drug scales as:
Cost per approval = Development cost ÷ Probability of success
When probability of success doubles from ~10% to ~20%, expected cost per approval can decrease by ~40–50%.
Claim Sequence
- Problem: ~90% of drug targets fail due to weak human causal validation.
- Standard fix: GWAS-supported targets provide ~2× higher approval odds.
- Additional insight: Extreme phenotypes and genotype-phenotype mismatch can enrich high-impact causal variants.
- Impact: 1.5–2.6× higher successful target identification in enrichment-oriented workflows.
- Outcome: Lower attrition, higher PoS, and substantial reduction in expected cost per approved drug.
Figure 1: Probability of Success
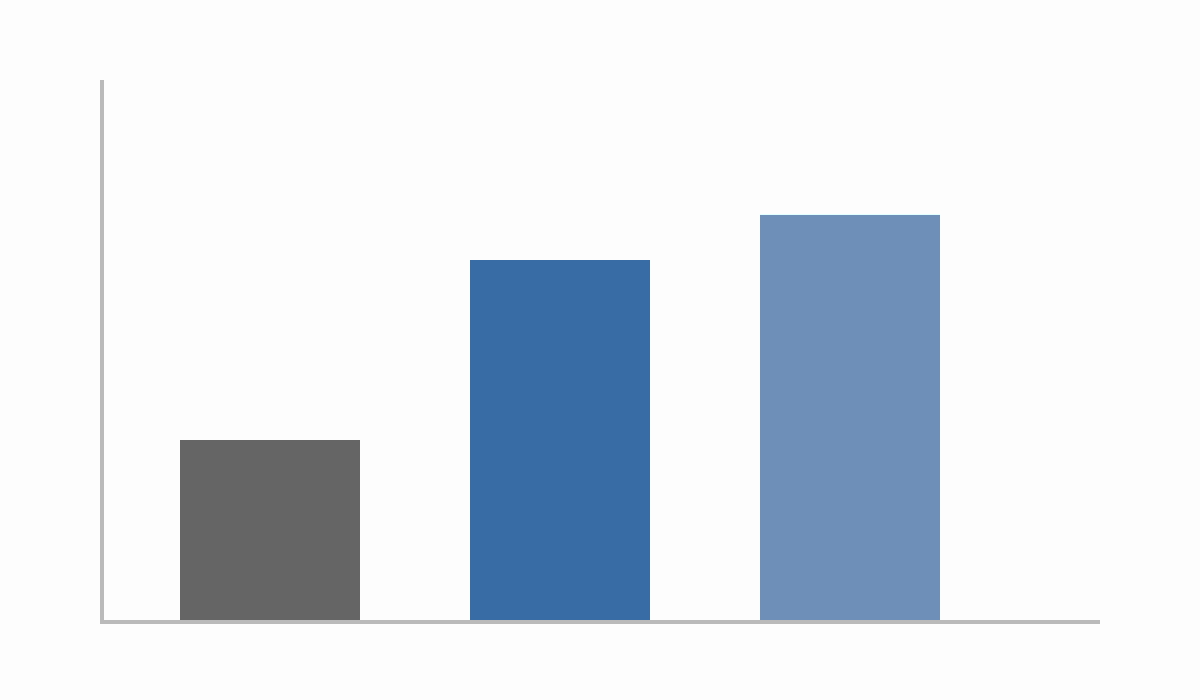
Figure 2: Cost Impact
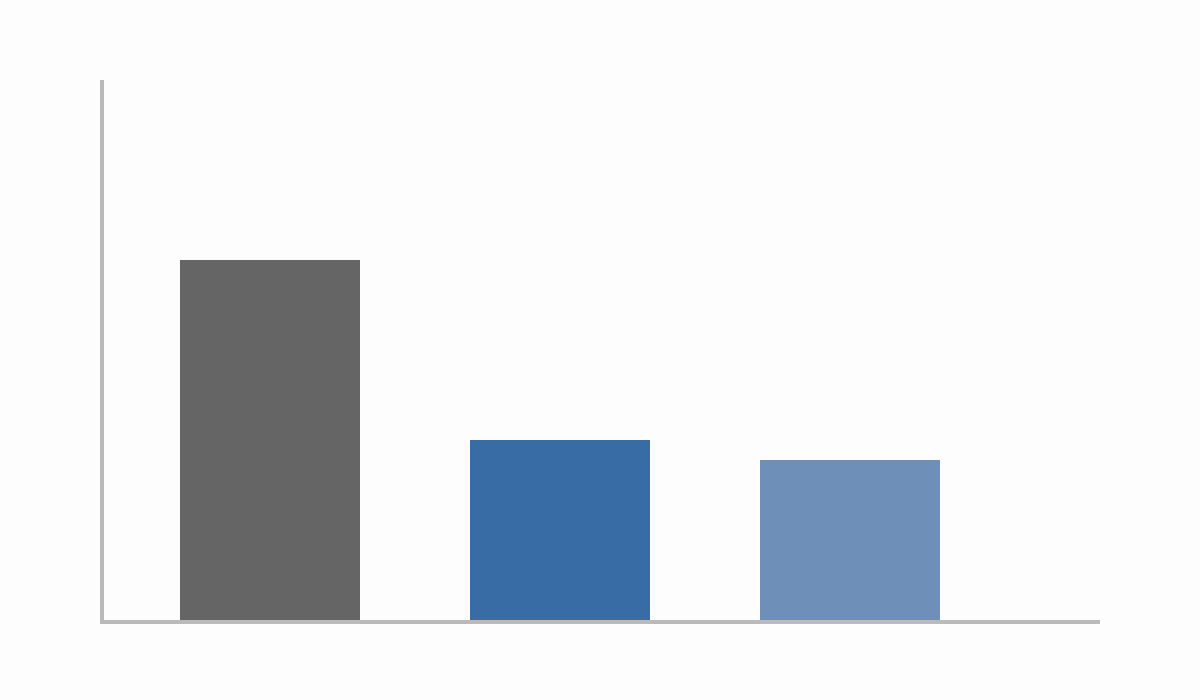
Compared with traditional non-genetic discovery, standard genetics can yield ≥40% lower expected R&D cost per approval. Enrichment-based genetics strategy can approach ~55% reduction, potentially representing over $1B savings per approved drug in some scenarios.